A Comparative Analysis of the Effects of Oral Gabapentin and Intravenous Dexamethasone on Postoperative Pain Following Hysterectomy: A Randomized Clinical Trial
- Department of Operating Room and Anesthesiology, School of Nursing and Midwifery, Zanjan University of Medical Sciences, Zanjan, Iran
- Department of Obstetrics and Gynecology, School of Medicine, Zanjan University of Medical Sciences, Zanjan, Iran
- Department of Nursing and Midwifery, School of Nursing and Midwifery, Shahid Beheshti University of Medical Sciences, Tehran, Iran
Abstract
Background: Optimizing post-operative pain management is paramount for patient recovery and satisfaction. Non-opioid analgesic agents, such as gabapentin and dexamethasone, are frequently utilized as adjunctive therapies to attenuate opioid consumption and mitigate associated adverse effects. This study sought to compare the analgesic efficacy of oral gabapentin versus intravenous dexamethasone administered pre-operatively in women undergoing elective abdominal hysterectomy.
Methods: In this randomized, single-blind clinical trial, 93 women scheduled for elective abdominal hysterectomy were randomly assigned to three cohorts (n = 31 each): oral gabapentin 600 mg, intravenous dexamethasone 8 mg, or a control group. Pain intensity was assessed using the Visual Analog Scale (VAS) at recovery and at 1, 6, 12, and 24 hours postoperatively. The requirement for supplemental analgesic (pethidine) intervention and postoperative complications were recorded. Data were analyzed using repeated measures ANOVA and Chi-square tests, with statistical significance defined as p < 0.05.
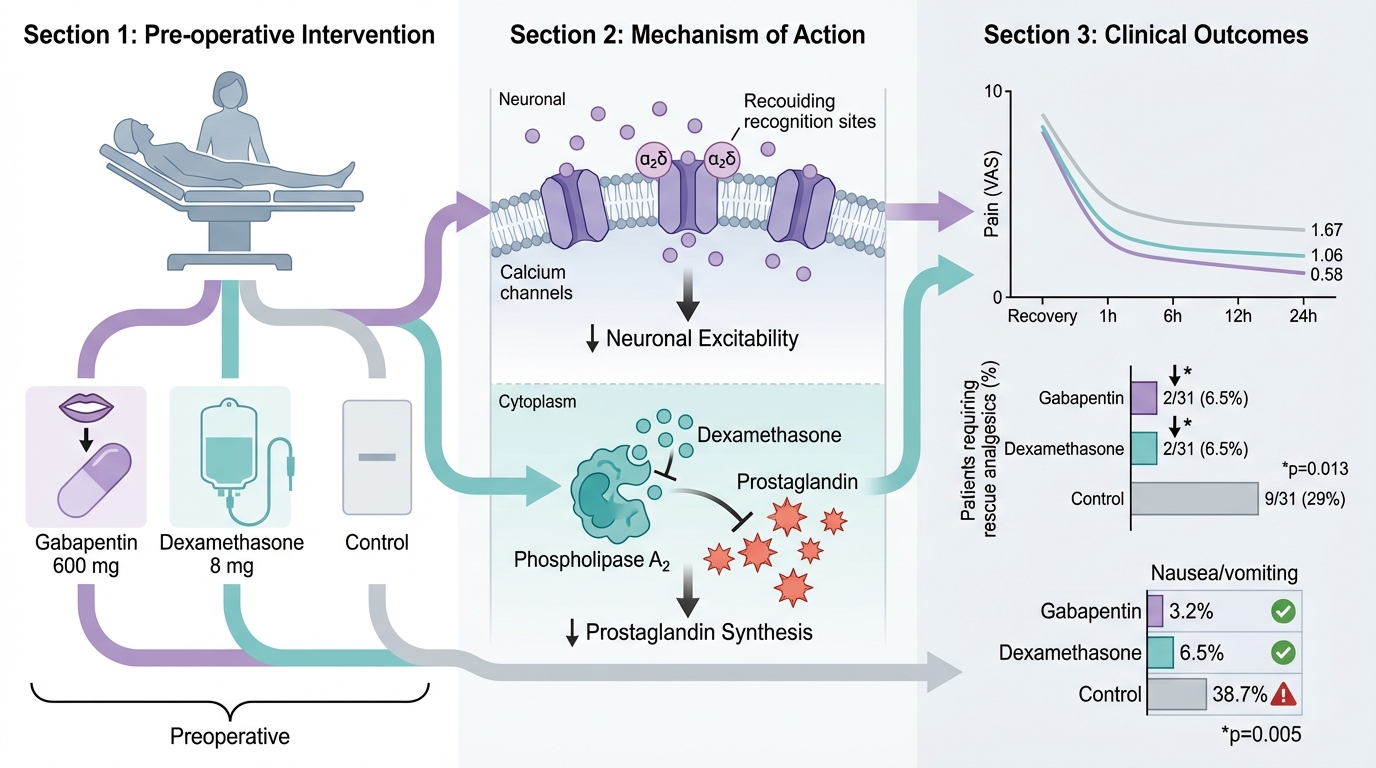
Results: Both the gabapentin and dexamethasone groups demonstrated significantly lower pain scores compared to the control group at all time points (p < 0.001). No significant difference was observed between gabapentin and dexamethasone up to 12 hours postoperatively ; however, at 24 hours, gabapentin provided lower pain scores (nominal p = 0.029; this difference did not persist after Bonferroni correction). The need for rescue analgesics and the incidence of nausea/vomiting were significantly lower in the intervention groups compared to the control group (p = 0.013 and p = 0.005, respectively). No serious adverse events were noted.
Conclusion: Pre-operative administration of oral gabapentin or intravenous dexamethasone effectively reduces post-operative pain and opioid requirements following hysterectomy. Although the difference at 24 hours did not meet the corrected threshold for statistical significance, gabapentin exhibited a trend toward more sustained analgesia, which may be attributable to its extended half-life. Both agents also mitigate postoperative nausea and vomiting, thereby enhancing patient comfort.
Introduction
Effective post-operative pain management continues to be a fundamental pillar of post-operative care, directly impacting recovery trajectories, length of hospital stay, patient satisfaction, and the incidence of complications. Suboptimal pain control may precipitate adverse physiological and psychological consequences, including delayed mobilization, diminished sleep quality, increased anxiety, and impaired nutritional intake, thereby placing an additional burden on healthcare systems 1. Accordingly, extensive efforts have been undertaken to minimize post-operative pain while concurrently reducing reliance on opioid analgesics. Although opioids remain the mainstay for acute pain relief, their adverse effects—such as respiratory depression, nausea, vomiting, pruritus, and ileus—have prompted the search for alternative pharmacological approaches 2.
One key strategy in modern pain modulation is pre-emptive analgesia, which involves the administration of analgesic agents prior to the onset of surgical nociception. This approach aims to prevent central sensitization to nociceptive stimuli and reduce the intensity of post-operative pain. Agents commonly employed in this strategy include opioids, non-steroidal anti-inflammatory drugs (NSAIDs), and NMDA receptor antagonists such as ketamine 3. In recent years, multimodal analgesia has emerged as a critical component of contemporary pain management. This concept entails the combination of analgesics with divergent mechanisms of action to enhance efficacy, attenuate side effects, and minimize opioid consumption. Clinical evidence supports the utility of such regimens in improving pain outcomes and patient satisfaction 4.
Among non-opioid options, gabapentin has gained considerable attention for its analgesic potential. Initially introduced in the 1990s as an antiepileptic agent 5 , gabapentin has since been employed in the management of neuropathic pain, anxiety disorders, social phobia, and restless leg syndrome 6. Its mechanism of action entails the modulation of neuronal excitability and the inhibition of neurotransmitter release—particularly glutamate—thereby attenuating peripheral and central sensitization 7. Gabapentin has demonstrated favorable results in reducing post-operative pain and opioid consumption, with a tolerable side effect profile comprising sedation, dizziness, dry mouth, and constipation 8,9,10. Its approximate 10-hour half-life facilitates effective dosing in perioperative settings.
Dexamethasone, a potent corticosteroid with anti-inflammatory and antiemetic properties, has also been incorporated into post-operative analgesic protocols. By inhibiting pro-inflammatory mediators such as prostaglandins, dexamethasone not only reduces pain but also significantly lowers the incidence of post-operative nausea and vomiting, particularly in patients undergoing general anesthesia 11. The selection of hysterectomy as the surgical model in this study is justified by its high prevalence and the variety of surgical techniques (abdominal, vaginal, and laparoscopic), which are often associated with substantial acute post-operative pain. Inadequate pain management following hysterectomy can impede recovery, increase opioid utilization, and reduce patient satisfaction 12. Although numerous studies have evaluated the analgesic efficacy of gabapentin and dexamethasone individually, comparative data on their preemptive use remain limited and inconclusive. Therefore, the present study was designed to compare the effectiveness of oral gabapentin versus intravenous dexamethasone, as non-opioid pre-medications, in modulating post-operative pain following hysterectomy.
Materials and Methods
Study Design
This study was a randomized, single-blind clinical trial.
Study Population
The study population comprised all women who were candidates for elective abdominal hysterectomy and were referred to Ayatollah Mousavi Hospital in Zanjan. The research was conducted in the operating room of Ayatollah Mousavi Hospital over an 11-month period at Zanjan University of Medical Science. The sample size was calculated based on the study by Forouzanfar et al. 13 . The calculation assumed an expected mean difference in Visual Analog Scale (VAS) scores of 1.15 with a common standard deviation of 0.25, a power of 90%, and an alpha level of 0.05 (two-sided). These parameters yielded a required sample size of 31 participants per group (total N=93), which was strictly adhered to in our study. Randomization was performed using balanced block randomization with a block size of three, resulting in 31 blocks and assigning 31 participants to each group. The study was single-blind; specifically, the patients were not blinded to their group allocation. However, the outcome assessors (research staff collecting the VAS scores) and data analysts were blinded to the group allocation throughout the study. Sampling was convenience-based and consecutive among women referred for elective hysterectomy in 2023.
Inclusion and Exclusion Criteria
The inclusion criteria were as follows: willingness to participate, ASA physical status class I or II, age between 30 and 65 years, candidate for elective abdominal hysterectomy due to benign indications, no known allergy to gabapentin or corticosteroids, no history of chronic pain or neurological/psychiatric disorders, no analgesic use in the past 24 hours, and absence of diabetes and hypertension. Exclusion criteria included withdrawal of consent at any time, surgery duration exceeding 3 hours, occurrence of any unusual intraoperative complications, and administration of analgesics during surgery.
Intervention groups
The study participants were randomly assigned to three groups: Group G received 600 mg oral gabapentin 30 minutes before surgery; Group D received 8 mg intravenous dexamethasone 30 minutes before surgery ; and Group C served as the control group and did not receive any pre-operative analgesic.
Procedure
After obtaining ethical approval from Zanjan University of Medical Sciences (IR.ZUMS.REC.1401.331) and registration in the Iranian Clinical Trials Registry, patients who provided informed consent completed the necessary forms. Demographic data and medical history—including age, comorbidities (diabetes, hypertension, cardiac diseases), and analgesic use—were recorded. All patients underwent a standardized general anesthetic protocol. Anesthesia was induced with intravenous midazolam (0.03 mg/kg), fentanyl (2 µg/kg), propofol (2 mg/kg), and atracurium (0.5 mg/kg) to facilitate endotracheal intubation. Anesthesia was sustained with isoflurane (1-1.5 MAC) in a 50% oxygen/air mixture, along with intermittent doses of fentanyl (1 µg/kg) and atracurium as needed based on the anesthesiologist's clinical assessment and monitoring of vital signs and depth of anesthesia.
Pain intensity was explained using the VAS, where 0 indicated no pain and 10 represented the worst imaginable pain. Pain was assessed by the researcher at recovery (T0), and at 1, 6, 12, and 24 hours post-operatively (T1–T4). If the VAS was ≥4, additional analgesia was provided with intravenous pethidine at a dose of 0.5 mg/kg, up to a maximum of 2 doses within 24 hours, and the total analgesic consumption was recorded. Diclofenac suppositories (100 mg) every 6 hours were administered to all patients following recovery. Potential drug-related adverse effects—such as hypotension, bradycardia, seizures, decreased consciousness, itching, skin rash, respiratory depression, nausea, and vomiting—were monitored and recorded during the initial 24 hours after anesthesia.
Statistical analysis
Data were entered into SPSS version 25. Descriptive statistics included means and standard deviations for quantitative variables and frequencies and percentages for qualitative variables. Normality was assessed using the Kolmogorov–Smirnov test. One-way ANOVA was used to compare means among groups. Qualitative variables were compared using the Chi-square test. For repeated measures of pain intensity over time, repeated measures ANOVA was utilized. A Bonferroni correction for multiple comparisons was applied, and a p-value < 0.0167 was considered statistically significant for these pairwise tests. A post-hoc power analysis, based on the observed effect sizes, confirmed that the study maintained adequate power (>90%) for the primary outcome.
Results
In this randomized clinical trial, 93 women scheduled for elective hysterectomy at Ayatollah Mousavi Hospital in Zanjan in 2023 and 2024 were enrolled and randomly assigned into three equal groups (n=31 each): Group G (oral gabapentin), Group D (intravenous dexamethasone), and Group C (control group with no premedication).
The mean age of participants was 46.08 ± 10.79 years (median: 45 years; range: 30–64 years). The average height was 163.45 ± 8.81 cm (median: 162 cm; range: 150–180 cm), and the mean weight was 69.39 ± 12.42 kg (median: 69 kg; range: 50–90 kg). Based on ASA classification, 47 patients (50.5%) were in class I and 46 patients (49.5%) were in class II. Statistical analysis revealed no significant differences among the three groups regarding age, height, weight, or ASA class, indicating demographic homogeneity (p > 0.05 for all variables) (
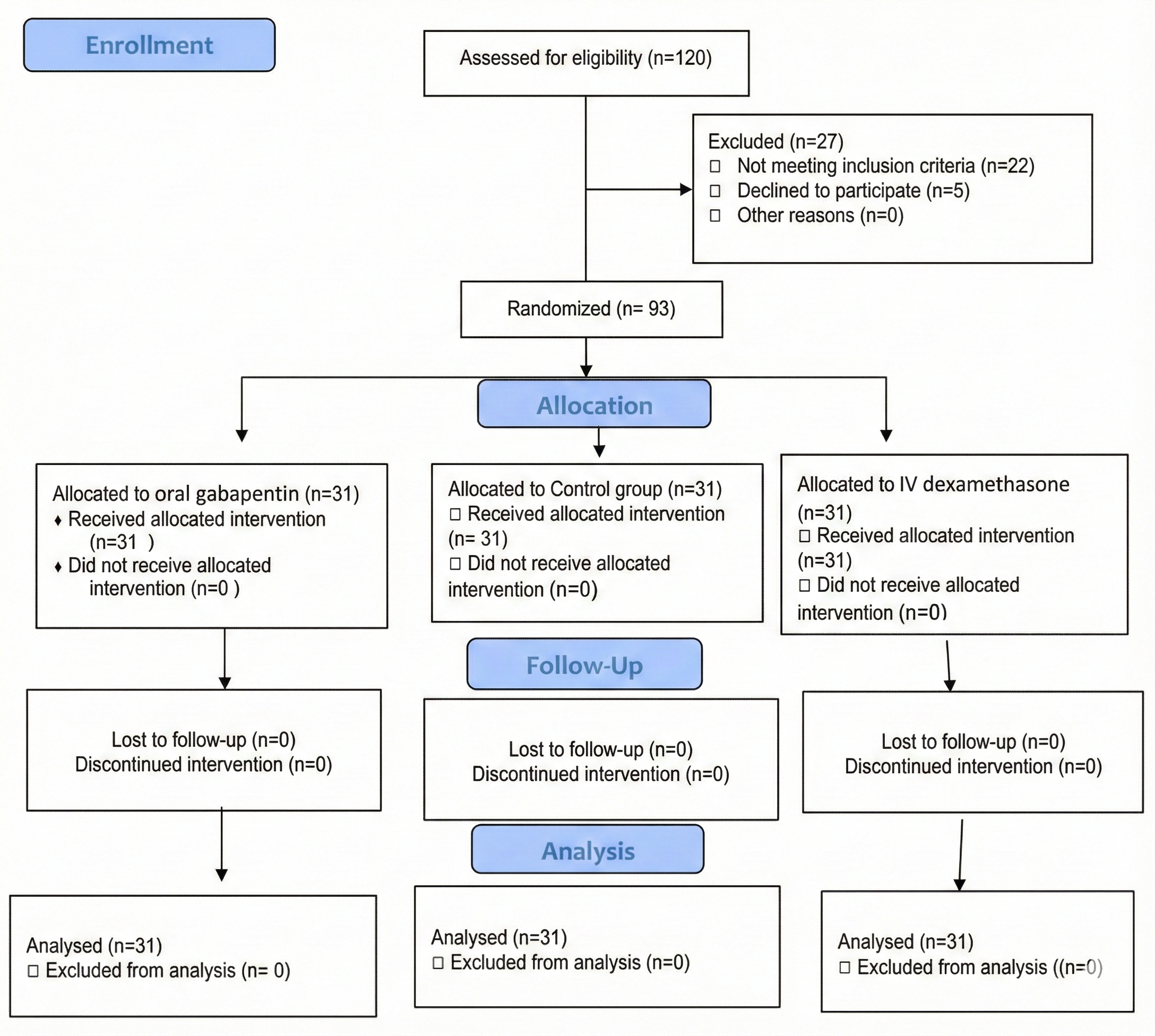
CONSORT Flow Diagram of Participant Screening, Randomization, and Follow-up
Demographic characteristics of patients in the three study groups
| Variable | Group G (n=31) | Group D (n=31) | Group C (n=31) | p-value |
|---|---|---|---|---|
| Age (years) | 46.35 ± 10.92 | 45.06 ± 9.83 | 46.83 ± 11.82 | 0.803 |
| Height (cm) | 165.12 ± 9.94 | 165.32 ± 6.94 | 161.90 ± 9.24 | 0.356 |
| Weight (kg) | 68.61 ± 13.39 | 72.00 ± 11.95 | 67.58 ± 11.81 | 0.345 |
| ASA-Class I | 15 (48.4%) | 16 (51.6%) | 16 (51.6%) | 0.958 |
| ASA-Class II | 16 (51.6%) | 15 (48.4%) | 15 (48.4%) |
The mean pain intensity (VAS) was documented at recovery and at 1, 6, 12, and 24 hours post-operatively. Comparison using repeated measures ANOVA showed a significant interaction between time and group (p < 0.001). Pain scores were significantly lower in Groups G and D compared to Group C at both the recovery time-point and 24 hours post-operatively. The overall time effect was also significant (p < 0.001), reflecting a consistent diminution in pain over time across all groups (
Pain intensity according to the VAS score over time among the three study groups
| Time point | Group G (n=31) | Group D (n=31) | Group C (n=31) |
|---|---|---|---|
| Recovery | 5.48 ± 0.50 | 5.61 ± 0.49 | 6.45 ± 0.50 |
| 1 hour | 4.51 ± 0.50 | 4.29 ± 0.46 | 6.29 ± 0.73 |
| 6 hours | 4.09 ± 0.83 | 4.48 ± 0.50 | 5.80 ± 0.94 |
| 12 hours | 3.54 ± 0.50 | 3.70 ± 0.82 | 3.87 ± 0.76 |
| 24 hours | 0.58 ± 0.50 | 1.06 ± 0.77 | 1.67 ± 0.87 |
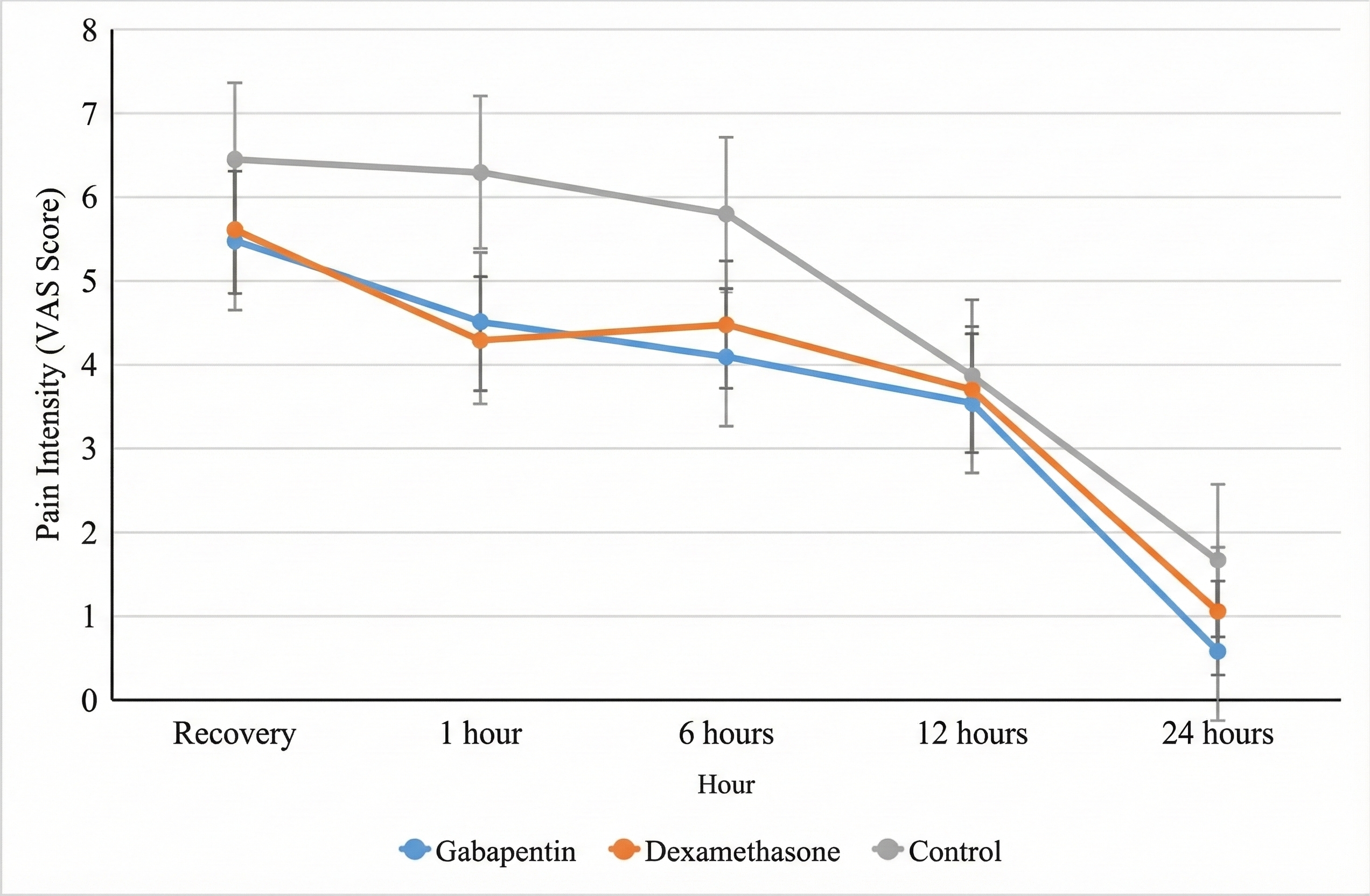
Comparison of Postoperative Pain Scores (Visual Analog Scale, VAS) Among the Three Study Groups Over Time
As shown in
Comparison of VAS Scores between groups (p-values)
| Time-point | Group G vs. Group D | Group G vs. Group C | Group D vs. Group C |
|---|---|---|---|
| Recovery | 0.573 | <0.001* | <0.001* |
| 1 hour | 0.283 | <0.001* | <0.001* |
| 6 hours | 0.132 | <0.001* | <0.001* |
| 12 hours | 0.646 | 0.180 | 0.646 |
| 24 hours | 0.029 | <0.001* | 0.004* |
The frequency of post-operative complications is presented in
Post-operative complications among the three study groups
| Complication | Group G (n=31) | Group D (n=31) | Group C (n=31) | Total (n=93) | p-value |
|---|---|---|---|---|---|
| Hypotension | 2 (6.5%) | 1 (3.2%) | 4 (12.9%) | 7 (7.5%) | 0.339 |
| Hypertension | 1 (3.2%) | 1 (3.2%) | 2 (6.5%) | 4 (4.3%) | 0.770 |
| Bradycardia | 0 (0%) | 2 (6.5%) | 2 (6.5%) | 4 (4.3%) | 0.118 |
| Tachycardia | 1 (3.2%) | 0 (0%) | 3 (9.7%) | 4 (4.3%) | 0.161 |
| Seizure | 0 | 0 | 0 | 0 | - |
| Impaired consciousness | 0 | 0 | 1 (3.2%) | 1 (1.1%) | 0.364 |
| Pruritus | 1 (3.2%) | 0 | 3 (9.7%) | 4 (4.3%) | 0.161 |
| Apnea | 0 | 0 | 0 | 0 | - |
| Nausea/Vomiting | 1 (3.2%) | 2 (6.5%) | 12 (38.7%) | 15 (16.1%) | 0.005 |
The need for additional analgesic (pethidine) administration post-operatively was significantly higher in the control group compared to the gabapentin and dexamethasone groups (p = 0.013). Only 2 patients (6.5%) in each of the G and D groups required pethidine, while 9 patients (29%) in the control group received rescue analgesia (
Need for additional analgesic (Pethidine) among the three study groups
| Requirement | Group G (n=31) | Group D (n=31) | Group C (n=31) | Total (n=93) | p-value |
|---|---|---|---|---|---|
| Yes | 2 (6.5%) | 2 (6.5%) | 9 (29.0%) | 13 (14.0%) | 0.013 |
| No | 29 (93.5%) | 29 (93.5%) | 22 (71.0%) | 80 (86.0%) |
Discussion
The present study was conducted to compare the efficacy of oral gabapentin (600 mg) and intravenous dexamethasone (8 mg) as non-opioid pre-medications in reducing post-operative pain following hysterectomy. The findings indicated that both agents markedly attenuated pain intensity and the need for rescue analgesics within the first 24 hours after surgery compared to the control group. Additionally, the incidence of nausea and vomiting was notably reduced in the intervention groups, highlighting the multifarious benefits of these pre-medications.
The analgesic mechanisms of these two agents, although distinct, are physiologically complementary. Gabapentin exerts its effect by binding to the α2δ subunits of voltage-gated calcium channels in the dorsal root ganglia, thereby reducing central neuronal excitability and attenuating hyperalgesia precipitated by inflammation or surgical injury 14. It also appears to reduce post-operative nausea through the modulation of neurotransmitters such as tachykinin 15,16. In contrast, dexamethasone reduces pain via the inhibition of phospholipase A2 and the cyclooxygenase and lipoxygenase pathways, leading to diminished synthesis of prostaglandins and neuropeptide release. Although its antiemetic mechanism remains partially obscure, it is hypothesized to involve central inhibition of prostaglandin synthesis and the enhancement of endogenous opioid release 10,14.
These results are consistent with prior investigations. Turan et al. 15 observed substantial pain reduction with 1200 mg gabapentin, albeit with higher rates of nausea and vomiting. Durmus et al. 17 demonstrated that gabapentin, alone or combined with acetaminophen, significantly reduced morphine consumption after hysterectomy. Similar findings were observed in studies by Dierking 9 and Fassoulaki 18, which confirmed the analgesic efficacy of pre-operative gabapentin in reducing post-operative pain and opioid use. Regarding dexamethasone, studies by Sugathan et al. and Srivastava et al. 6,19 have shown its effectiveness in alleviating both pain and nausea after laparoscopic hysterectomy.
When interpreting these findings, it is essential to consider not only statistical significance but also clinical relevance. Although the nominal pain score difference between the gabapentin and dexamethasone groups at 24 hours was notable (p=0.029), this finding did not remain statistically significant after applying the Bonferroni correction for multiple comparisons. Its clinical significance therefore necessitates careful consideration. The ultimate value of an intervention is determined by its ability to provide a perceptible benefit to the patient. Therefore, while both drugs are effective, the choice between them should be guided by a holistic view that includes the clinical importance of the observed differences alongside their side effect profiles and cost.
A notable aspect of the current study is the direct comparison between two non-opioid agents using standardized single doses and different administration routes. While both agents showed comparable analgesic effects up to 12 hours post-operatively, gabapentin demonstrated superior pain control at the 24-hour mark, likely due to its protracted half-life. This suggests that although both agents are effective in the acute post-operative period, gabapentin may offer a more enduring analgesic effect.
Despite its methodological strengths, the study possesses several limitations. First, as patients were aware of their group allocation (receiving an oral medication, an intravenous injection, or no premedication), this may have introduced expectation bias into their self-reporting of pain scores (VAS). While we attempted to mitigate this by employing blinded outcome assessors, this remains an important limitation when interpreting the subjective pain outcomes. Secondly, the study population was limited to women undergoing elective hysterectomy at a single academic center, which restricts the generalizability of the findings to other settings or populations. Additionally, the single-center design may introduce environmental or procedural biases. The follow-up period was confined to 24 hours post-operatively, precluding assessment of long-term outcomes such as chronic pain or delayed complications. Moreover, the use of pethidine as the rescue analgesic, while reflecting our institutional protocol, is a limitation as its neurotoxic metabolite profile makes it less desirable compared to alternative modern opioids. Furthermore, important patient-centered outcomes, such as sleep quality, satisfaction, and hospital stay duration, were not evaluated. Finally, the investigation was limited to a fixed dose of each drug, precluding the assessment of dose-response relationships.
Conclusion
Based on the findings of this study, pre-operative administration of oral gabapentin (600 mg) or intravenous dexamethasone (8 mg) approximately 30 minutes prior to elective hysterectomy appears to be safe and effective in significantly reducing post-operative pain intensity and the requirement for additional opioid analgesics during the first 24 hours after surgery. Although the difference at 24 hours did not meet the corrected threshold for statistical significance, gabapentin showed a trend toward more sustained analgesia, which may be attributable to its extended half-life.
Abbreviations
ANOVA: Analysis of Variance; ASA: American Society of Anesthesiologists; NMDA: N-Methyl-D-Aspartate; NSAIDs: Non-Steroidal Anti-Inflammatory Drugs; PONV: Post-Operative Nausea and Vomiting; SPSS: Statistical Package for the Social Sciences; VAS: Visual Analog Scale
Acknowledgments
This research was approved and financially supported by the Deputy of Research and Technology at Zanjan University of Medical Sciences. The authors would like to express their sincere appreciation to the medical staff at Mousavi Hospital for their valuable assistance throughout the study.
Authors’ contributions
Vahideh Rashtchi: Conceptualization, Methodology, Investigation, Data Curation, Writing - Original Draft, Project Administration. Hamideh Gholami: Conceptualization, Methodology, Resources, Writing - Review & Editing, Supervision, Project Administration, Formal Analysis. Neda Zamanpour: Investigation, Data Curation, Writing - Review & Editing. Helia Fallah Yari: Data Curation, Writing - Review & Editing.
Funding
This study was supported by the Deputy of Research and Technology at Zanjan University of Medical Sciences. The funder had no role in the design of the study, data collection, analysis, interpretation of data, or writing of the manuscript.
Availability of data and materials
The datasets generated and analyzed during the current study are not publicly available due to patient confidentiality constraints but are available from the corresponding author upon reasonable request.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Zanjan University of Medical Sciences (Approval Code: IR.ZUMS.REC.1401.331). Informed written consent was obtained from all individual participants included in the study.
Consent for publication
Not applicable.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work, the authors used Deep Seek in order to improve readability and language. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Competing interests
The authors declare no competing interests.

